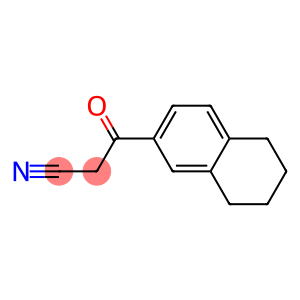
3-(1,2,3,4-Tetrahydronaphthalen-6-yl)-3-oxopropanenitrile, 3-(5,6,7,8-Tetrahydro-2-naphthyl)-3-oxopropionitrile
(5,6,7,8-四氢-2-萘甲酰基)乙腈
CAS: 2623-25-8
Molecular Formula: C13H13NO
3-(1,2,3,4-Tetrahydronaphthalen-6-yl)-3-oxopropanenitrile, 3-(5,6,7,8-Tetrahydro-2-naphthyl)-3-oxopropionitrile - Names and Identifiers
3-(1,2,3,4-Tetrahydronaphthalen-6-yl)-3-oxopropanenitrile, 3-(5,6,7,8-Tetrahydro-2-naphthyl)-3-oxopropionitrile - Physico-chemical Properties
| Molecular Formula | C13H13NO |
| Molar Mass | 199.25 |
| Melting Point | 93-100 °C |
3-(1,2,3,4-Tetrahydronaphthalen-6-yl)-3-oxopropanenitrile, 3-(5,6,7,8-Tetrahydro-2-naphthyl)-3-oxopropionitrile - Risk and Safety
| Hazard Symbols | Xn - Harmful |
| Risk Codes | 22 - Harmful if swallowed |
| WGK Germany | 3 |
3-(1,2,3,4-Tetrahydronaphthalen-6-yl)-3-oxopropanenitrile, 3-(5,6,7,8-Tetrahydro-2-naphthyl)-3-oxopropionitrile - Introduction
(5,6, 7,8-Tetrahydro-2-naphthoyl) acetonitrile ((5,6,7,) methylene)acetonitrile) is an organic compound. The following is a detailed description of the properties, uses, preparation and safety information of the compound:
1. nature:
(5,6, 7,8-tetrahydro-2-naphthoyl) acetonitrile is a colorless liquid with an aromatic odor. Its molecular formula is C12H13N and its molar mass is 171.24g/mol. It has a melting point of -8°C and a boiling point of 235°C. It is soluble in organic solvents such as ethanol and methylene chloride at normal temperature, and hardly soluble in water.
2. use:
(5,6, 7,8-tetrahydro-2-naphthoyl) acetonitrile is an important organic synthesis intermediate. It is often used in series reactions, formylation reactions, reduction reactions and group exchange reactions in organic synthesis reactions. It is also widely used in drug synthesis, pesticide synthesis and fragrance synthesis.
3. preparation method:
The preparation of (5,6, 7,8-tetrahydro-2-naphthoyl) acetonitrile can be carried out by the following steps:
a. An acylation reaction is performed on naphthalene and formaldehyde to obtain a naphthoyl compound.
B. The obtained naphthoyl compound is reacted with acetonitrile under basic conditions to give the final product (5,6, 7,8-tetrahydro-2-naphthoyl) acetonitrile.
4. Safety Information:
(5,6, 7,8-tetrahydro-2-naphthoyl) acetonitrile is generally less hazardous under normal use and storage conditions. However, when handling this substance, the following precautions still need to be paid attention:
a. Avoid contact with skin and eyes, and wear appropriate protective equipment when using.
B. avoid inhalation of its vapor or dust, try to operate in a well-ventilated place.
c. The substance should be stored in a cool, dry place, away from fire and oxidant.
d. please observe the relevant safety operation procedures and precautions during use.
Please note that the above is only a brief introduction. For more detailed and accurate information, it is recommended to consult the relevant scientific literature and chemical manuals.
1. nature:
(5,6, 7,8-tetrahydro-2-naphthoyl) acetonitrile is a colorless liquid with an aromatic odor. Its molecular formula is C12H13N and its molar mass is 171.24g/mol. It has a melting point of -8°C and a boiling point of 235°C. It is soluble in organic solvents such as ethanol and methylene chloride at normal temperature, and hardly soluble in water.
2. use:
(5,6, 7,8-tetrahydro-2-naphthoyl) acetonitrile is an important organic synthesis intermediate. It is often used in series reactions, formylation reactions, reduction reactions and group exchange reactions in organic synthesis reactions. It is also widely used in drug synthesis, pesticide synthesis and fragrance synthesis.
3. preparation method:
The preparation of (5,6, 7,8-tetrahydro-2-naphthoyl) acetonitrile can be carried out by the following steps:
a. An acylation reaction is performed on naphthalene and formaldehyde to obtain a naphthoyl compound.
B. The obtained naphthoyl compound is reacted with acetonitrile under basic conditions to give the final product (5,6, 7,8-tetrahydro-2-naphthoyl) acetonitrile.
4. Safety Information:
(5,6, 7,8-tetrahydro-2-naphthoyl) acetonitrile is generally less hazardous under normal use and storage conditions. However, when handling this substance, the following precautions still need to be paid attention:
a. Avoid contact with skin and eyes, and wear appropriate protective equipment when using.
B. avoid inhalation of its vapor or dust, try to operate in a well-ventilated place.
c. The substance should be stored in a cool, dry place, away from fire and oxidant.
d. please observe the relevant safety operation procedures and precautions during use.
Please note that the above is only a brief introduction. For more detailed and accurate information, it is recommended to consult the relevant scientific literature and chemical manuals.
Last Update:2024-04-09 20:13:35